DFT Study: Geometry Optimization and Electronic Properties of Steroid Derivative Compounds
DOI:
https://doi.org/10.51574/hayyan.v3i1.3878Keywords:
DFT; HOMO-LUMO; MEPAbstract
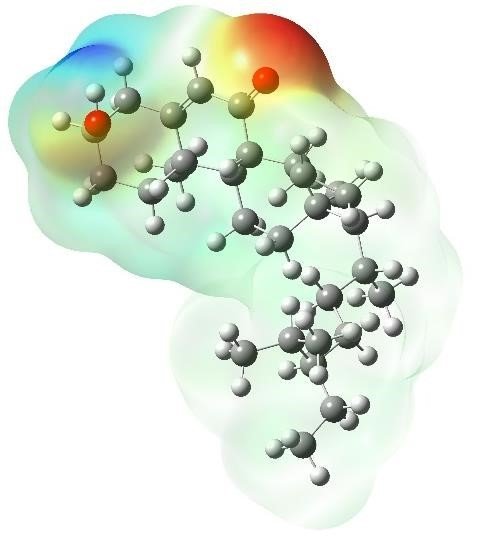
This study presents a descriptive investigation of three steroidal compounds—6-hydroxystigmast-4-en-3-one (1), stigmast-4-en-3-one (2), and 3-hydroxystigmast-5-en-7-one (3)—using Density Functional Theory (DFT) at the B3LYP level with the 6-31G**(d,p) basis set. The calculations were performed to analyze the optimized geometries and electronic properties of the compounds. The presence of hydroxyl and carbonyl groups within these molecular structures revealed their roles as electron-donating sites and potential active centers for electrophilic attack. Furthermore, several quantum chemical descriptors, including chemical hardness (δ), softness (σ), electronegativity (χ), chemical potential (µ), and electrophilicity index (ω), were determined to provide insights into the reactivity and stability of the studied molecules.
References
Borah, P. & Banik, B. K. Diverse synthesis of medicinally active steroids. Green Approaches in Medicinal Chemistry for Sustainable Drug Design (Elsevier Inc., 2020).
Indriani, I., Aminah, N. S. & Puspaningsih, N. N. T. Antiplasmodial Activity of Stigmastane Steroids from Dryobalanops oblongifolia Stem Bark. Open Chem. 18, 259–264 (2020).
Banabic, D. et al. Hosford WF and Caddell RM (1995) Metal Forming-Mechanics and Metallurgy. 2nd edn. Prentice-Hall. Kocks UF, Tó me CN and Wenk HR (1998) Texture and Aniso-tropy. Int. J. Plast. 4, 339 (2000).
Tandon, H., Chakraborty, T. & Suhag, V. A Brief Review on Importance of DFT In Drug Design. Res. Med. Eng. Sci. 7, 791–795 (2019).
Deghady, A. M., Hussein, R. K., Alhamzani, A. G. & Mera, A. Article density functional theory and molecular docking investigations of the chemical and antibacterial activities for 1-(4-hydroxyphenyl)-3-phenylprop-2-en-1-one. Molecules 26, (2021).
Xu, Y., Chu, Q., Chen, D. & Fuentes, A. HOMO–LUMO Gaps and Molecular Structures of Polycyclic Aromatic Hydrocarbons in Soot Formation. Front. Mech. Eng. 7, 1–10 (2021).
Drissi, M. et al. Theoretical and experimental electrostatic potential around the mnitrophenol molecule. Molecules 20, 4042–4054 (2015).
Downloads
Published
Versions
- 2026-02-28 (2)
- 2026-02-28 (1)

